Best Of The Best Info About How To Draw Molecular Geometry
Determine the number of lewis electrons.
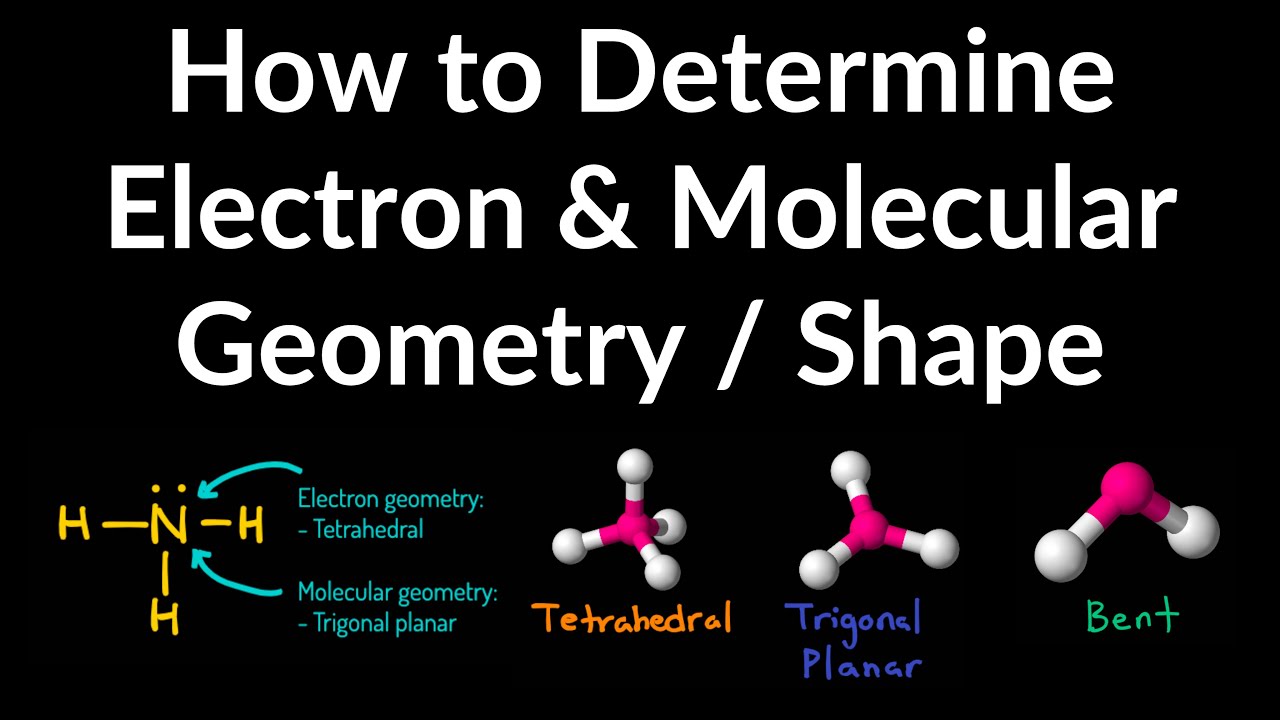
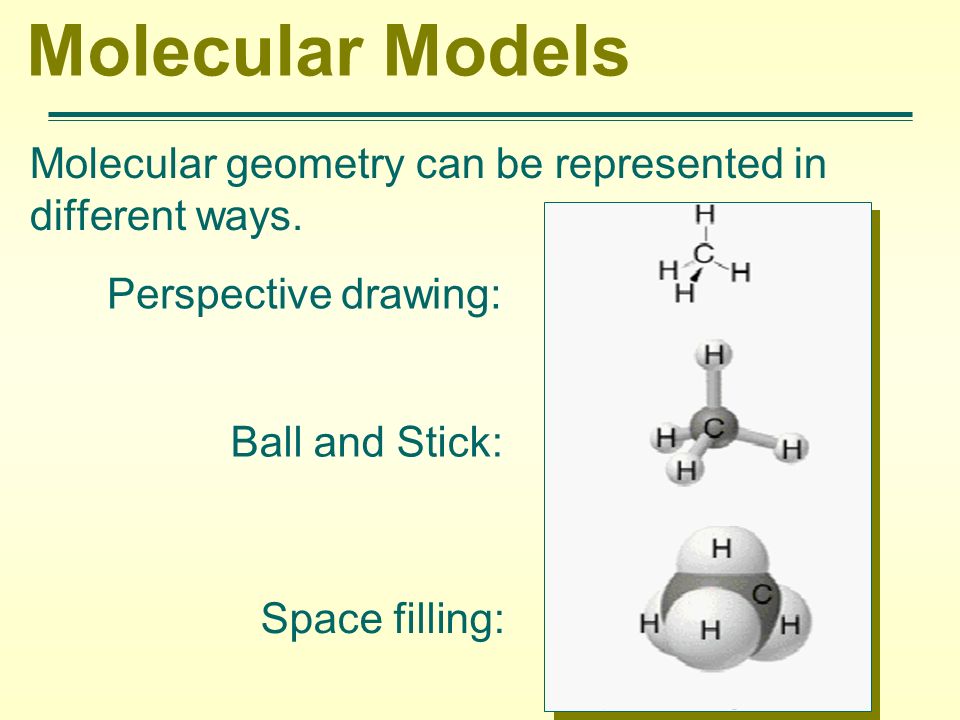
How to draw molecular geometry. Two examples of how to determine molecular geometry, bond angle, hybridization, and polarity.helpful resources:• how to draw lewis structures: This depends on the atoms in the molecule. This video show how draw a simple molecule (i.e., phthalic acid) using gaussview software.
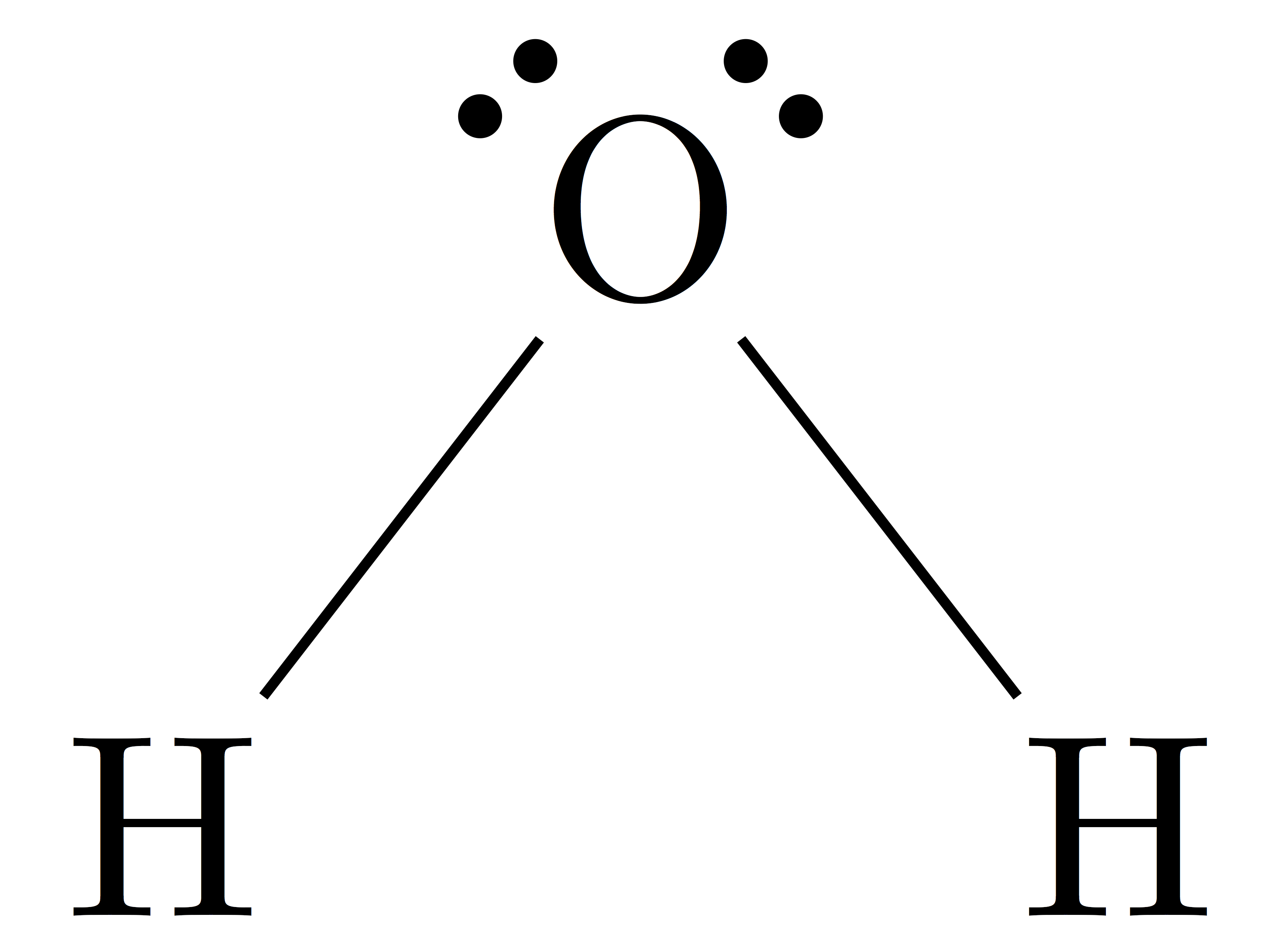
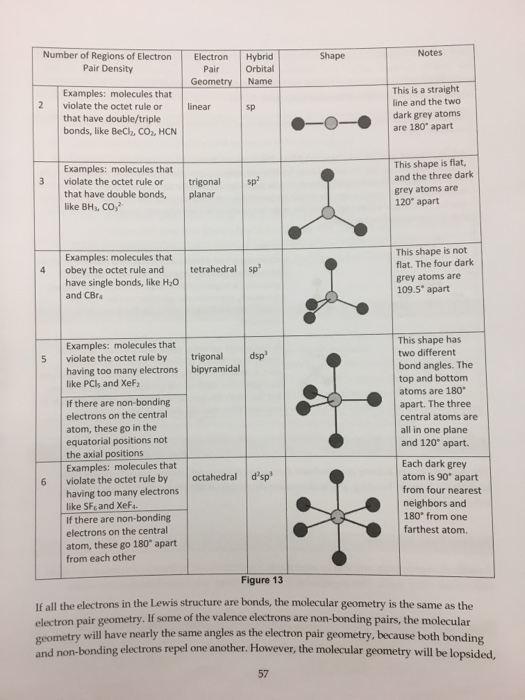
To chose the central atom as the one with the smallest number. How to draw a lewis structure. The first step is to sketch the molecular geometry of.
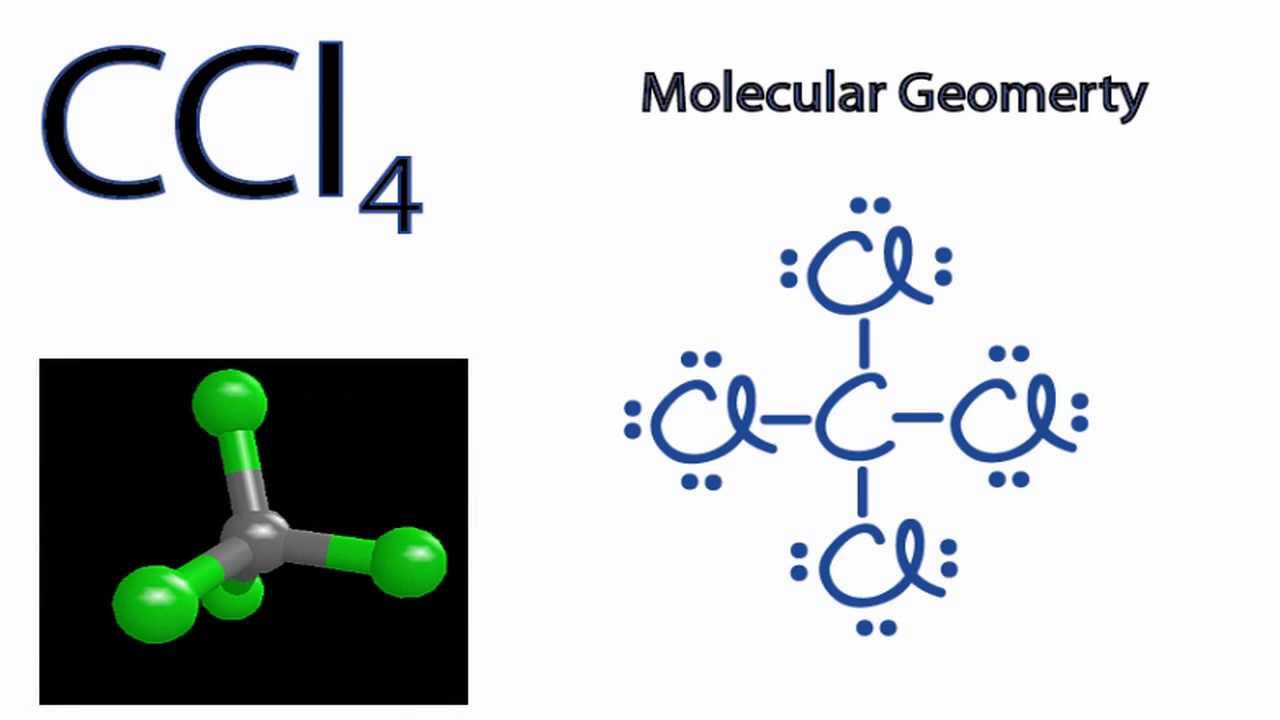
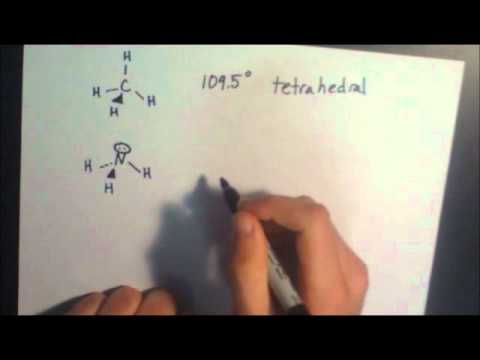
Next, count the total number of electron groups on the central atom. The first step is to draw the lewis structure of the molecule. 6 draw bonds (shared pairs) from the central atom to each surrounding.
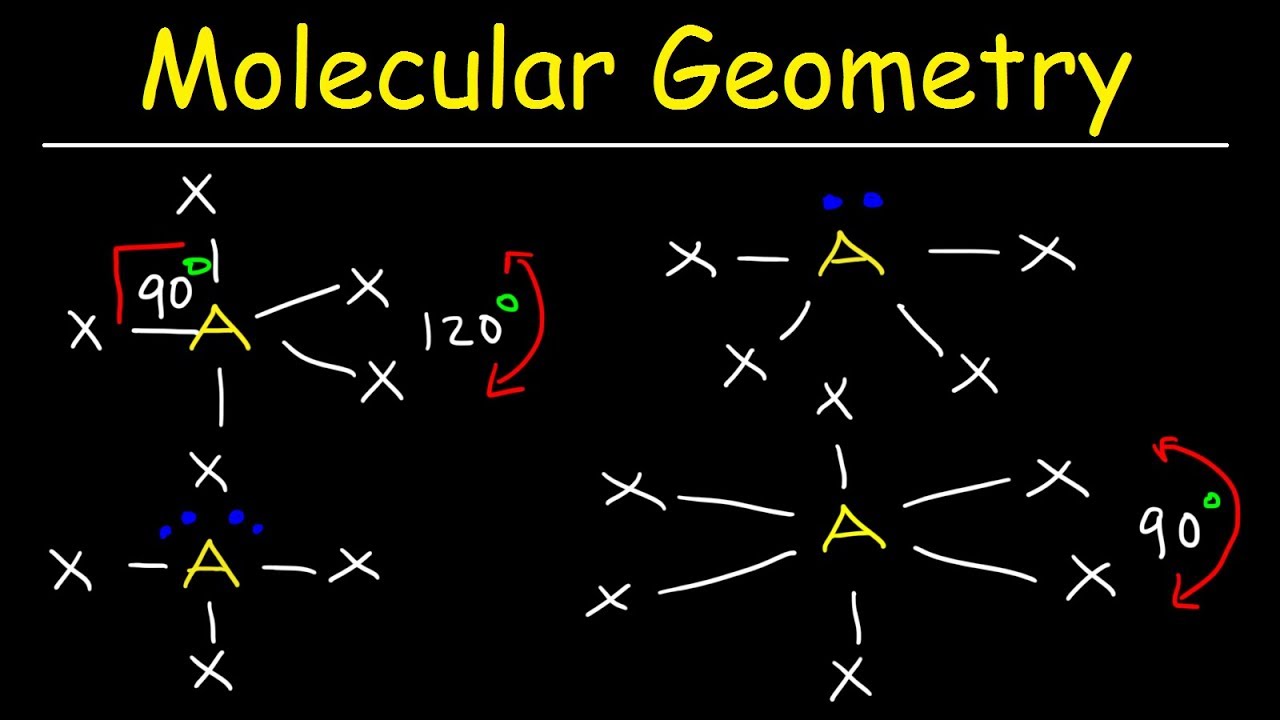
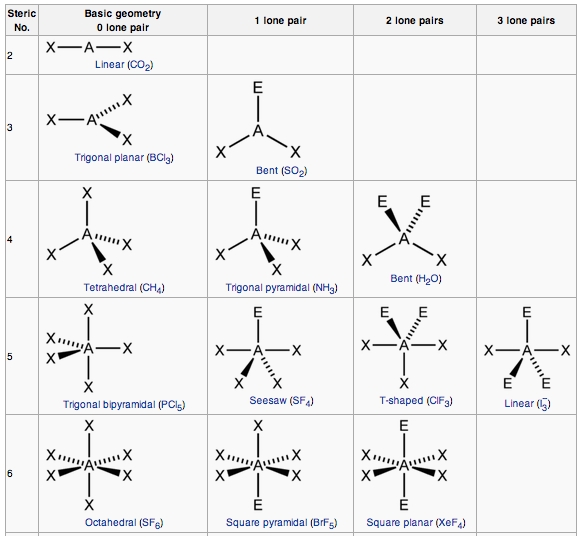
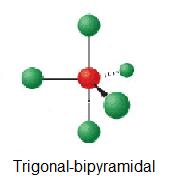
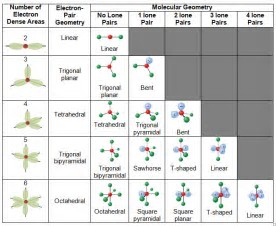
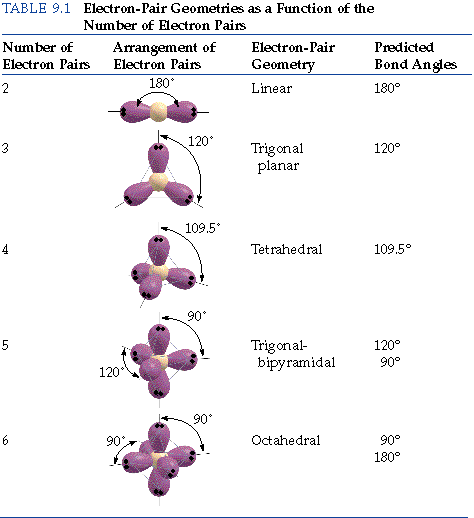
Around phosphorus, there are four electron groups: There are two fundamental principles at play here: Molecular geometries (linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral) are determined by the vsepr theory.
Draw the molecule using a lewis diagram. 14 rows how to use the table to predict molecular geometry. The molecular geometry can be.
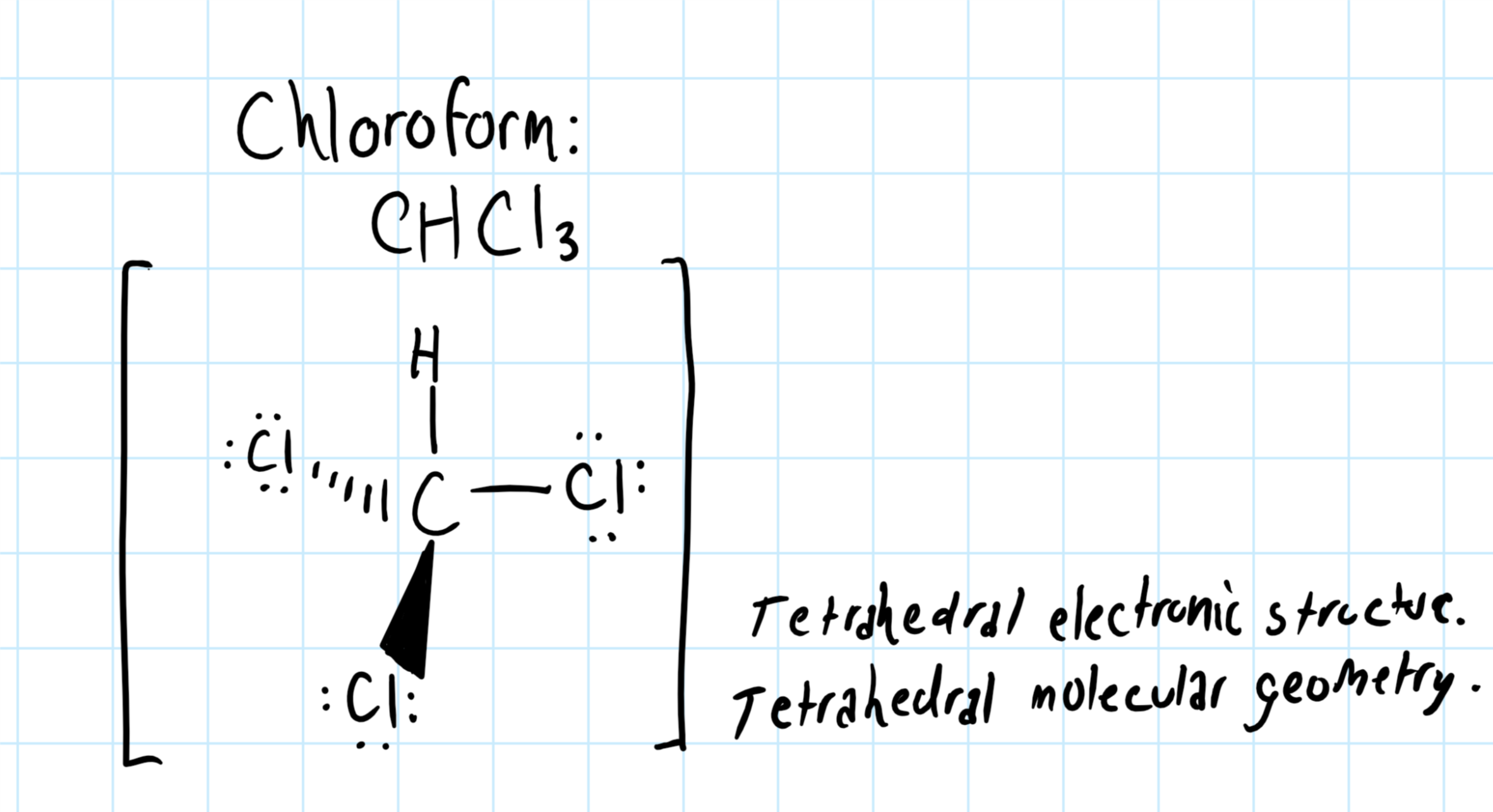
Also, the molecule was submited to gaussian 09w for optimization w. To predict the shape of a covalent molecule, follow these steps: The lewis diagram of a molecule (nh3) can be drawn by following the step below;
Ketzbook explains molecular geometry, vsepr theory, and the 5 basic shapes of molecules with examples for each one. Count the total number of valence electrons in a molecule. In this video we’ll use vspre theory to practice the rules for identifying the major molecular geometries, including bond angles.
A table of geometries using the vsepr theory can facilitate. The first step is to sketch the molecular geometry of. The valence electrons of nitrogen.
Watch to the end to learn how you can ge.